电解质及隔膜研究
在锂离子电池中,阴极和阳极通常由含有液体电解质的隔膜分离。另一方面,固体电解质正处于研发阶段,其不但在安全性及能量密度方面进行了有意义的提升,同时也节约了成本。
液体电解质
最常用的液体电解质是锂盐溶液(LiPF6或LiBF4)中添加粘合剂和添加剂,粘合剂和添加剂的加入增加粘度并减少气体逸散。液体电解质在充放电过程中会发生降解并释放气体,这会导致电池膨胀,同时某些反应产物还会导致电池腐蚀,如分解产物氢氟酸。
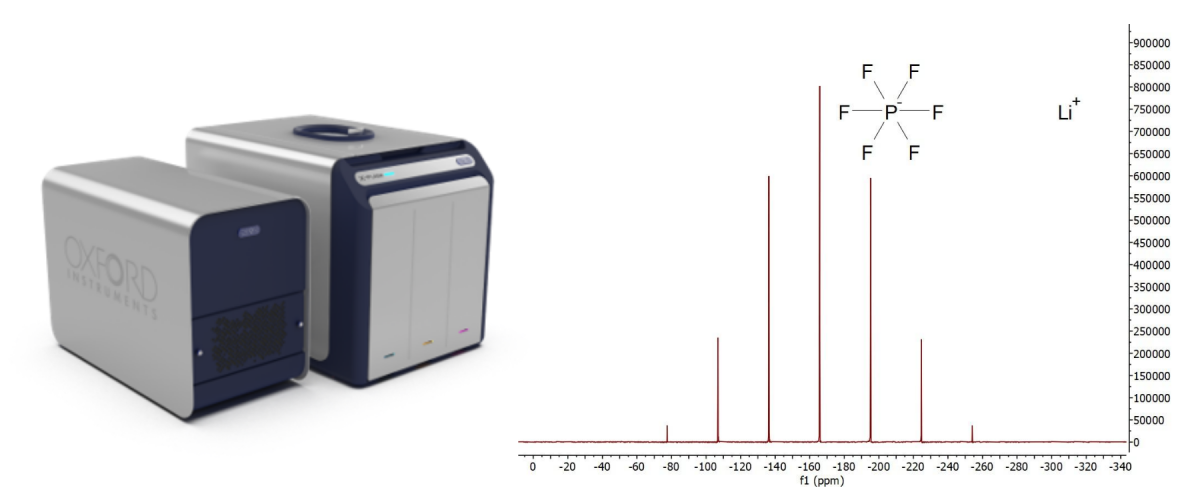
牛津仪器无制冷剂X-Pulse核磁共振系统是唯一能在一个系统中完成所有成分(1H,13C, 19F,7Li,31P,11B)测量的台式核磁共振波谱仪。仪器不但能准确识别和量化HF和LiF等分解产物,同时还可测量扩散系数。
图1(右): X-Pulse核磁共振系统能够在现场条件下进行全电解液表征或质量控制。电解质LiPF6反应产物PF6的核磁谱图。

固体电解质
人们研究了多种可作为锂离子电池固体电解质的材料。这些材料可以使用金属锂做负极,比现有的电池更安全,潜在的能量密度更高。然而,锂离子在固态电解液中的电导率通常低于液态电解质。通过能谱(EDS)获得的Li元素面分布图,可以提供有关材料结构以及任何有害枝晶形成的重要信息。与电导率测量相结合,可有效推动固态电解质性能的研究,并进一步促使全固态电池成为可能。Ultim Extreme能谱仪可以在足够低的束流下检测锂元素,同时不会明显改变样品。
图2:(左)采集前的固态石榴石电解质;(中)利用 Ultim Extreme在2.5k, 100pA条件下采集的元素面分布图——确定了三种不同的含锂相;(右)采集后相同的区域,几乎无损伤
相关产品
 点击回看
点击回看

 公安机关备案号31010402003473
公安机关备案号31010402003473